Ethylene oxide (EtO) is an essential component of modern medical device manufacturing and is frequently used in sterilization processes as one of the most effective methods for mitigating the growth of microorganisms on devices used by doctors and patients every day. However, even as EtO allows for the mass production of critical, live-saving medical devices, other hazards exist from exposure to this toxic gas.
While EtO is a listed hazardous air pollutant by the EPA, source and fugitive emissions limitations in the current federal rules have not been able to satisfy the scrutiny imposed by many state environmental agencies and community groups. Following the 2016 IRIS risk assessment by the EPA, which classified EtO as more toxic than previously understood, many concerned stakeholders began investigating EtO emissions sources that may be contributing to exposure in communities. This resulted in a growing number of lawsuits and legal implications for medical device sterilizers along with a push by the EPA and many states to tighten the regulatory requirements. According to the FDA, approximately 55% of all medical devices in the U.S. are sterilized with EtO which creates a significant level of complexity for implementing regulations around a chemical necessary to maintain critical supply chains.
By working with commercial sterilization facilities worldwide, Picarro and its partners are currently assisting with a wide range of monitoring solutions to prepare for this evolving regulatory landscape and enabling companies to generate the defensible data they need.

Partnering for Innovative Ethylene Oxide Solutions
Assessing the Risk of Sterilization Operations
In the United States, each facility’s emissions limitations have primarily been defined through air permitting and applicable regulation under the Clean Air Act (CAA). 40 CFR 63 Subpart-O, the National Emissions Standards for Hazardous Air Pollutants (NESHAP) specific to commercial sterilization and fumigation operations initially adopted in 1994, establishes the emissions requirements at facilities using greater than 1-ton of EtO per year. In order to further evaluate the effectiveness of this current rule, the EPA published an advance notice of proposed rule-making (ANPR) for comment and information requests on December 12, 2020. As the EPA considers extending the Toxics Release Inventory (TRI) reporting requirements, a number of information collection requests have followed including the expanded section 313 notice to additional sterilization facilities based on proximity to population areas and emissions estimates. In a more aggressive approach, many states have taken action to regulate EtO from source and facility-associated fugitive emissions with requirements far more stringent than the current NESHAP. Some states have implemented site-specific requirements by placing process throughput limitations on EtO and increasing destruction and capture efficiency standards for abatement control equipment. Other agencies have developed specific conditions for preventing fugitive emissions' promulgation by mandating sterilizers to operate under negative pressure zones (i.e. EPA method 204) covering areas where off-gassing of sterilized product may occur. The mechanism for these requirements has ranged from state-enforced consent decrees, permitting, and even bills passed through the legislature. While these are examples of state environmental agency requirements, this evolving regulatory landscape also provides perspective on the expectations that may be implemented during the upcoming NESHAP revision.
Now is an excellent time for commercial sterilization facilities to assess the trend of current and expected regulatory requirements and implement a dynamic strategy that engages key stakeholders. By reviewing current monitoring technology and site-specific system requirements, companies can optimize sterilization and emissions control processes and improve employee safety. With a team of experts dedicated to providing monitoring solutions for facilities using EtO, Picarro can assist with assessing and effectively mitigating risk related to indoor monitoring (mobile or multi-point), continuous emissions monitoring systems (CEMS), and fence-line or near-source monitoring systems.
Minimizing Employee Exposure by Upgrading Indoor Monitoring Systems
Research around fugitive emissions and growing concern with employee workplace exposure has driven many commercial sterilization facilities to challenge the validity of their antiquated indoor monitoring systems. These systems have traditionally been comprised of a multi-point monitoring network utilizing electrochemical, metal oxide sensors (MOS), Fourier-transform infrared spectroscopy (FTIR), and Gas chromatography (GC) based PID or FID analyzers. Unfortunately, EHS and industrial hygiene staff reliant on the poor performance and slow detection speed of these older systems are finding the data to be unreliable. As a result, companies are required to primarily rely on passive monitoring programs which are not intended to promote early detection of high EtO concentration exposure but rather an integrated average over a specified time period.
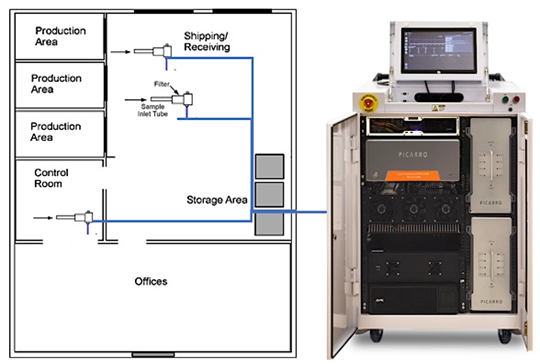
Figure 1: Picarro Multi-Point Indoor Monitoring System for Maximum Spatial Coverage in a Commercial Sterilization Facility
With traditional systems failing to meet the monitoring needs of industrial facilities and passive monitoring methods preventing a proactive approach, there is a clear need for innovative technologies. Picarro has developed an ideal solution to meet these very demands of facilities looking for a prudent methodology for upgrading indoor air monitoring programs that will promote employee safety, early detection of leaks or process issues, and defensible data to accurately quantify fugitive emissions. Aging sensors and monitoring systems often display higher than actual indoor EtO concentrations due to highly susceptible interferences from other hydrocarbons and water vapor. Now, with analyzers tuned specifically for measuring EtO at sub-ppb levels with an industry-leading time resolution, Picarro-based systems address the monitoring needs of single and multi-point monitoring configurations with the lowest operational cost (Figure 1).
Ensure Compliance with Continuous Emissions Monitoring Systems (CEMS)
Following the revised risk value of EtO, reducing emissions from industrial sources has become a strong focus of the EPA and many state agencies. Increased regulation targeted at lowering emissions has already taken effect in certain states with federal rule-making expected to follow. This regulatory landscape has driven advances in abatement control technology beyond 99% efficiency such as that developed by Solsteo, a service provider that designs, manufactures, and installs sterilization vessels and control equipment for medical sterilizers. Facilities are being challenged to control and monitor emissions with the most precise and sensitive technology available. Traditional CEMS technologies such as optically enhanced FTIR and GC are inherently susceptible to interferences and fail to quantify EtO at the very low concentrations required for future compliance with emissions limitations. In addition, the requirement for CEMS is already being implemented by some states and is expected to be a condition in the commercial sterilizer NESHAP 40 CFR 63 subpart-O rule update in 2022.
With this expectation of future CEMS requirements, Picarro’s solution with EtO-specific Cavity Ring-Down Spectroscopy (CRDS) analyzers offer the precision, speed, and sensitivity levels necessary to comply with future regulations. As shown in Figure 2, Picarro has partnered with CleanAir Engineering, providing a turn-key approach to allow for real-time insights in continuous flow and concentration levels on one or more final exhaust stacks. Automated reporting, simplified operational training, notifications for early detection, mass emissions rate reporting, and less downtime allow CEMS customers to operate reliable monitoring solutions to mitigate the risk of non-compliance.
Managing the Narrative Through Continuous Fence-line and Near-Source Monitoring
Commercial sterilization facilities are often faced with significant difficulty in navigating the growing public scrutiny surrounding EtO. Over the past few years, state regulatory changes have involved some previously unfamiliar requirements for sterilizers such as fence-line ambient air monitoring. Currently, Illinois is the only state to formalize a fence-line monitoring requirement for its facilities using EtO, but similar federal legislation has been proposed by Senator Tammy Duckworth (Ill.) and Representative Lisa Blunt Rochester (Del.). This requirement was introduced in the Public Health Air Quality Act of 2020, which later failed to receive a vote. This is a clear indication that environmental-related actions and legislation are often introduced as a result of demand by community members concerned with the implications that industrial activity may have on their health and well-being.
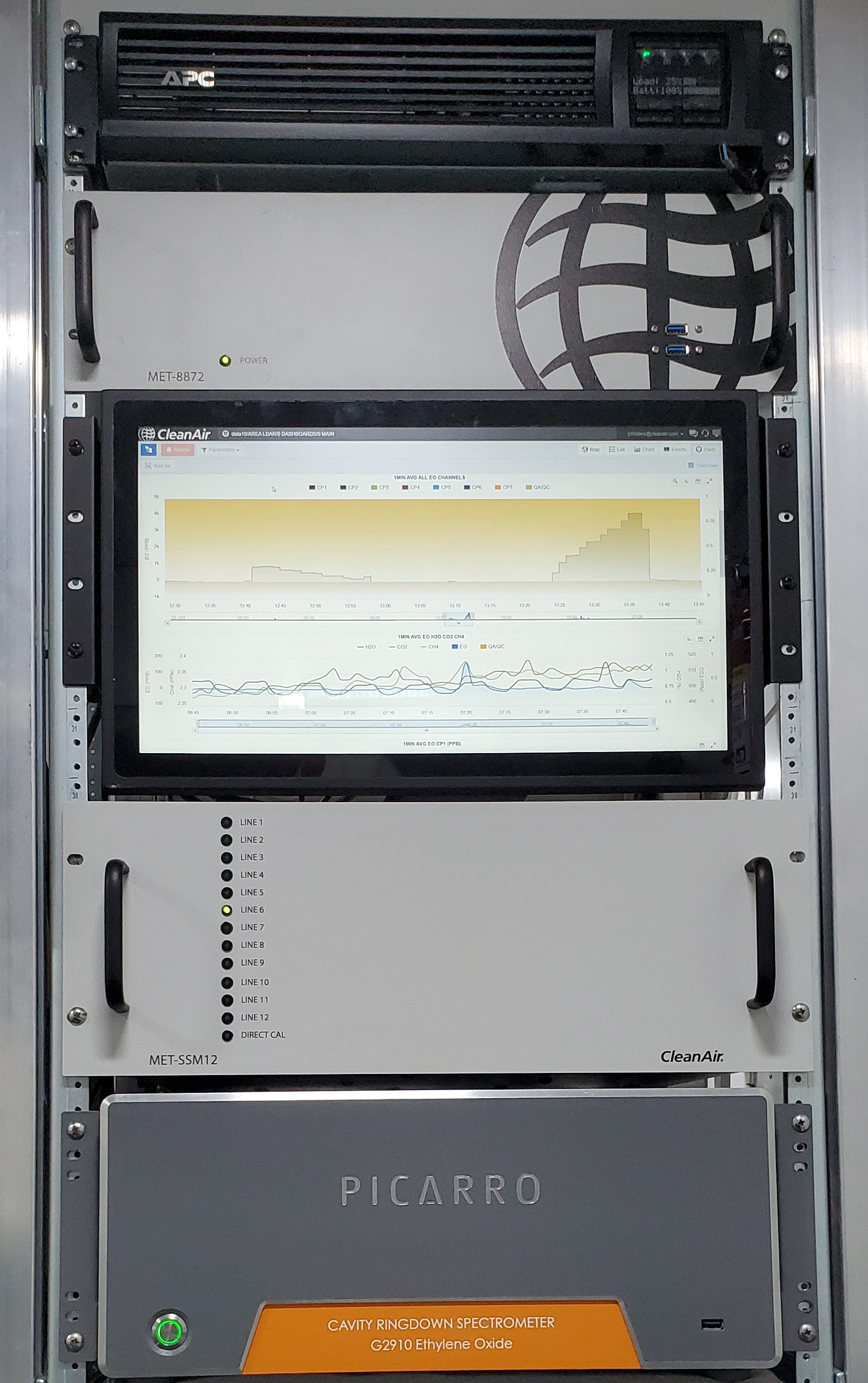
Figure 2: CleanAir Engineering Implementation of a Picarro-based Ethylene Oxide Air Quality Monitoring System.
Fence-line monitoring is typically completed through a passive canister sampling methodology found in TO-15, the EPA toxic organics ambient air sampling method. However, as identified by the EPA and many consultants performing ambient air monitoring programs, significant inconsistencies exist with canister materials and lab testing equipment, often creating inflated EtO measurements. Along with positive bias created from the "canister effect," this methodology can only provide a time-averaged sample, which is later analyzed in a lab with mass spectrometry. Figure 3 depicts a Picarro instrument specific for operating in fence-line applications enabling facilities to generate real-time data that can also be correlated with meteorological conditions (i.e., wind direction) for air modeling and spatial analysis of EtO concentrations. This is a significant progression towards proactive mitigation of fugitive emissions and offsite impacts from EtO usage, further allowing companies to present a positive narrative of their efforts to the many concerned stakeholders.

Figure 3: Picarro’s G2920 EtO Analyzer Providing Continuous 2-Second Ambient Measurements with Best-in-Class Sensitivity for Fence-line Monitoring.
What's Next?
Commercial sterilizers are at acritical point with pending regulations potentially affecting all aspects of their operation. Part of the anticipated regulation will be the requirement to continuously monitor for EtO emissions at process vents and stacks. In addition, many facilities are required to maintain ambient air monitoring efforts for EtO as well as perform indoor air monitoring to mitigate employee exposure risks. Companies generating EtO emissions should take a pragmatic stance on evaluating and utilizing the best monitoring solutions for their facility needs. Assessing current technology utilized by regulatory agencies for their monitoring projects provides a direct approach to exploring available monitoring solutions. For example, the Georgia Environmental Protection Division (EPD) utilizes Picarro for an ambient EtO measurement study funded by the EPA. At the same time, the Houston Health Department acquired Picarro instruments to conduct real-time formaldehyde measurements along the river channel in a large industrial area. However, evaluating the core technical advantages of each technology provides essential insight needed when choosing the best monitoring systems. Picarro and their partners accomplish this in a variety of ways including application and technology review discussions with prospective customers as well as onsite demonstrations. With clear advantages in measurement speed, sensitivity, and reliability over traditional technologies, Picarro’s EtO analyzers provide a uniform solution to mitigate current and future regulatory risk with indoor, source, and fence-line measurements. Please visit the EtO product information page or contact us to learn more.

